The regulatory landscape of regenerative medicine worldwide continues to evolve as scientific advancements accelerate. Governments and health authorities must balance innovation with patient safety. As a result, regulatory frameworks differ significantly across regions, influencing how therapies reach the market.
Regenerative medicine includes stem cell therapy, tissue engineering, and biologics. Therefore, consistent oversight is essential to ensure ethical and clinical standards are met.
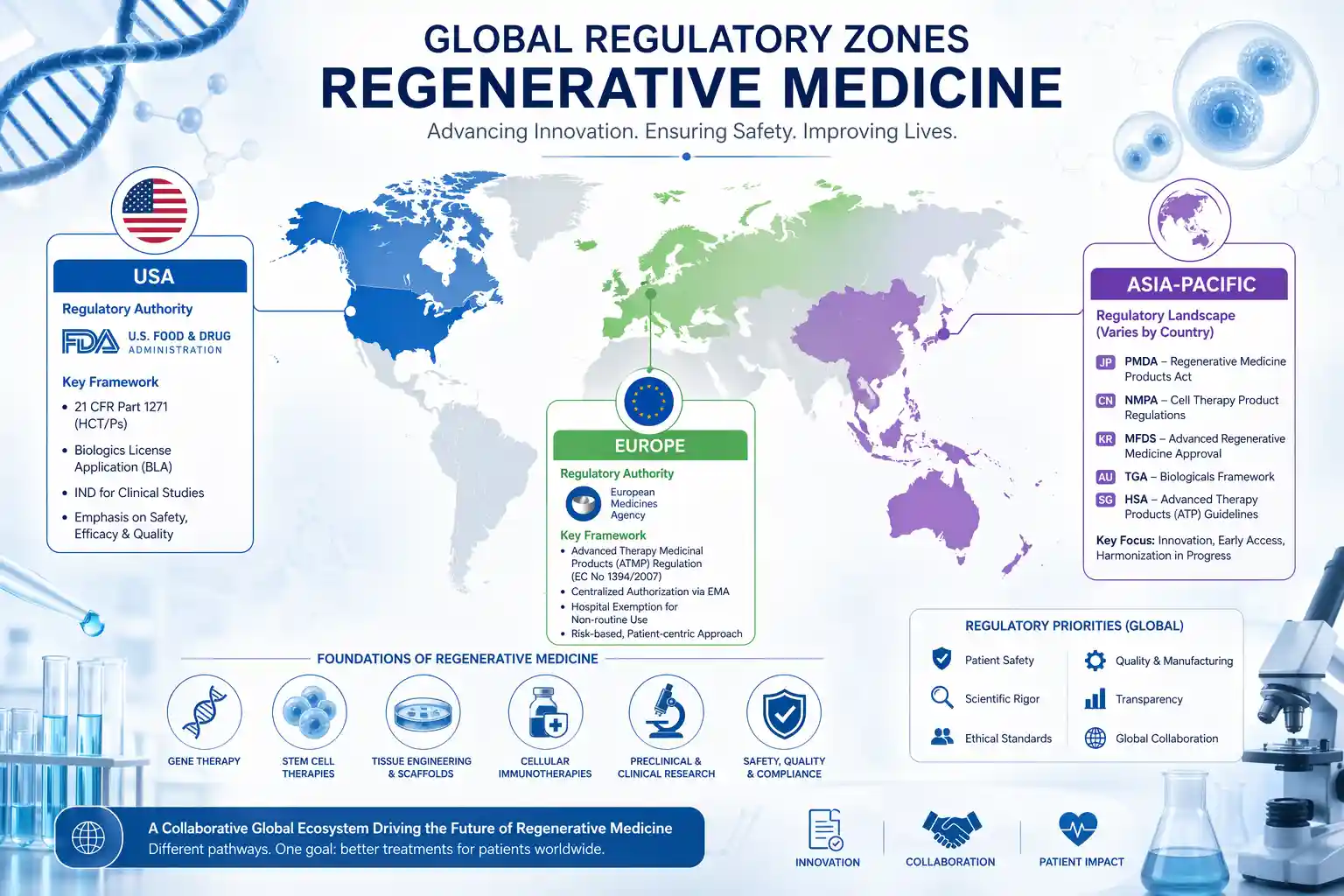
Several major regulatory bodies influence the regulatory landscape of regenerative medicine worldwide:
These organizations help define safety, efficacy, and manufacturing standards globally.
The United States applies a rigorous approval process. Clinical trials must demonstrate safety and efficacy before commercialization. This approach ensures reliability but may slow innovation.
The European Union uses centralized procedures for ATMPs. Although harmonized, individual countries may implement additional requirements.
Countries like Japan and South Korea adopt accelerated approval systems. These frameworks allow earlier patient access while ongoing data collection continues.
Such variations highlight how the regulatory landscape of regenerative medicine worldwide is shaped by regional priorities.
Despite progress, several challenges remain:
These issues complicate cross-border collaboration and patient safety.
For deeper insights, visit:
Proper training is critical for navigating the regulatory landscape of regenerative medicine worldwide. Clinicians must understand compliance requirements, documentation, and ethical considerations.
IARM Training provides structured programs to help professionals stay aligned with global standards. Learn more at: https://iarm-usa.com/
Regulatory systems are adapting to keep pace with innovation. Key trends include:
These developments will continue shaping the regulatory landscape of regenerative medicine worldwide.
The regulatory landscape of regenerative medicine worldwide is complex yet essential for ensuring safe and effective therapies. While regional differences exist, global collaboration is improving consistency. Moving forward, education, innovation, and regulation must work together to advance the field responsibly.
