Regenerative medicine is transforming how diseases and injuries are treated, but its real impact depends on one critical step: translation. Translational regenerative medicine bridges the gap between laboratory discoveries and safe, effective clinical therapies. For clinicians, researchers, and healthcare organizations, understanding this pathway is essential to advancing patient care while maintaining scientific and ethical rigor.
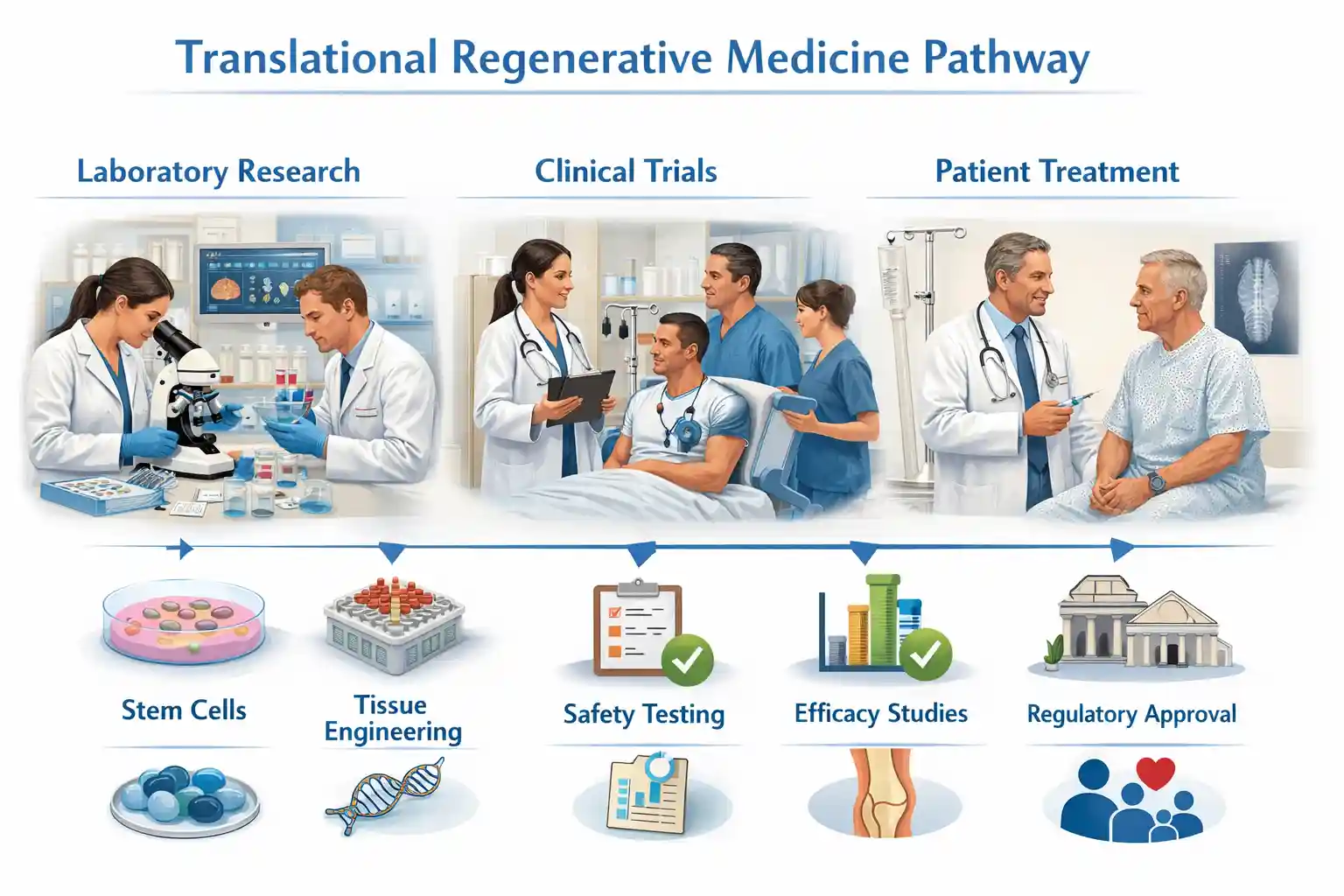
Translational regenerative medicine focuses on converting scientific breakthroughs—such as stem cell biology, tissue engineering, and biologics—into clinically viable therapies. While basic research uncovers mechanisms of healing, translation ensures these findings are applied responsibly in real-world medical settings.
Importantly, this process aligns innovation with patient safety. Therefore, regulatory frameworks, standardized protocols, and evidence-based practice play a central role.
At this stage, regenerative medicine research explores cellular behavior, biomaterials, and signaling pathways. These discoveries are validated through in vitro studies and animal models to assess feasibility and biological response.
Once safety is demonstrated, therapies enter phased clinical trials. Translational regenerative medicine emphasizes trial design, patient selection, and outcome measurement. As a result, treatments are refined before broader clinical adoption.
Standardization is essential. Good Manufacturing Practice (GMP) ensures consistency, sterility, and reproducibility of regenerative products. Without this step, clinical scalability would not be possible.
Finally, therapies are introduced into clinical practice through trained providers. Continuous monitoring, data collection, and post-market surveillance support long-term success and patient trust.
For healthcare professionals, translational regenerative medicine provides a framework for integrating innovation without compromising standards of care. It ensures that new therapies are:
Scientifically validated
Ethically applied
Compliant with regulatory expectations
Moreover, it empowers clinicians to confidently educate patients about realistic outcomes and risks.
Education is a key driver of successful translation. Organizations like International Association of Regenerative Medicine focus on equipping physicians with the knowledge needed to interpret research, evaluate therapies, and apply regenerative medicine responsibly.
Programs and resources available through https://iarm-usa.com/ support clinicians at every stage of professional development, from foundational science to advanced clinical application.
NIH – Translational Research Overview
https://www.nih.gov/research-training/medical-research-initiatives/clinical-translational-science-awards
FDA – Regenerative Medicine Advanced Therapies (RMAT)
https://www.fda.gov/vaccines-blood-biologics/cellular-gene-therapy-products/regenerative-medicine-advanced-therapy-designation
IARM Education and Training Programs
https://iarm-usa.com/
As regenerative medicine continues to evolve, translation will remain the defining factor between promise and practice. With proper training, regulation, and collaboration, translational regenerative medicine will continue to redefine patient-centered care and clinical outcomes worldwide.
